Chemotherapy: Biologics
Episode Notes
Figure 1: Receptor Tyrosine Kinases: tyrosine kinases are part of cell surface receptors that bind to growth factors. When growth factor binding is facilitated, this allows for over expression of cells/proliferation. Tyrosine kinases are appealing therapeutic targets as they are located on the cell surface. Non-receptor kinases are enzymes that phosphorylate proteins in the inner cell membrane and cytoplasm (non cell surface receptor). Include kinases that phosphorylate tyrosine residues of target proteins (SRC family) and specific kinases for serine/threonine residues (AKT family). Phosphatases (like PTEN) oppose activity of kinases), PTEN is a tumor suppressor which negatively regulates the PI3K signaling pathway.
Image from: https://www.nature.com/articles/s41392-022-01168-8
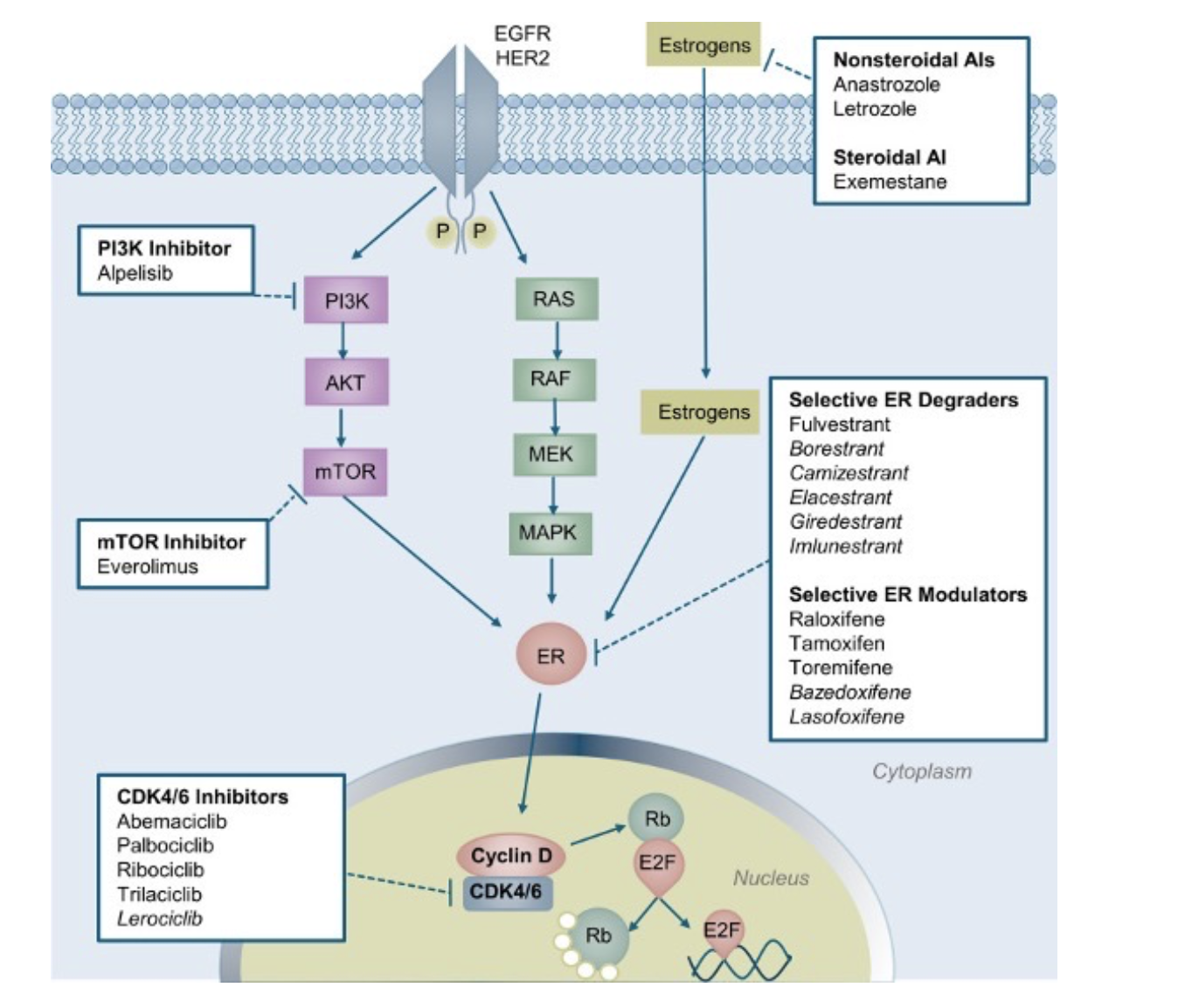
Figure 2: Pathways and Target Agents: Combining endocrine and targeted therapies to overcome resistance. Resistance to endocrine therapies is common in hormone-driven cancers. Current strategies to overcome this resistance are focused on inhibiting upstream signaling pathways and CDK4/6. Approved therapies, and those currently in clinical development (italicized), are depicted with mechanisms of action that include targeting estrogens, estrogen receptor (ER), growth signaling pathways, and CDK4/6.
Image from: https://www.sciencedirect.com/science/article/pii/S1048891X24014725
Antiangiogenic therapies
Bevacizumab: covered previously, see show notes from episode 15 - PARPi, Bev, and ovarian cytotoxics
Tyrosine kinase inhibitors: cell surface receptors which bind to growth factors > proliferation of cells
Most are multikinase inhibitors (i.e. not specific to a single receptor)
Pazopanib (multikinase)
No specific FDA approval in gyn cancers (but has NCCN guideline recommendations)
Oral administration (once daily, without food)
Common AE: fatigue, hypertension, diarrhea, elevated liver enzymes, nausea, vomiting, and abdominal pain
Less common severe AE:
Cardiotoxicity, with QT prolongation and risk of CHF
Hemorrhagic/thrombotic events
Impaired wound healing
GI perforation and fistulas
Other AE: ILD, PRES, hypothyroidism, proteinuria, infection, tumor lysis syndrome
Management/monitoring for AE
Hepatotoxicity: get liver enzymes at baseline, 2 weeks after starting treatment, repeat throughout treatment
Cardiotoxicity: EKG, electrolytes, echo at baseline and during treatment
Hold around time of surgery to avoid complications with hemorrhage, clotting, wound healing, fistula
Lenvatinib (multikinase)
FDA approval: in combination with pembro for the use in pMMR advanced/recurrence endometrial cancer (KEYNOTE-775/Study 309)
Oral administration (daily, swallow capsules whole, typical dose 20mg)
Dose reductions: down to 8mg/dose (seemingly preserves efficacy)
Can consider starting at a dose <20mg to reduce toxicity
AE (with len/pem together, as lenvatinib currently not used alone in gyn cancer)
89% of patients experience a grade 3+ event
5.7% rate of fatal adverse events in the treatment arm
>65% required dose reduction/interruption. 33% discontinued the drug.
Common: hypothyroidism, hypertension, fatigue, diarrhea, and MSK disorders, which all occur in >50% of patients who take this combination. N/V and stomatitis occur in >⅓ of all patients.
Monoclonal antibodies
Trastuzumab: MAB against HER2/ERBB2 (part of the EGFR tyrosine kinase family)
Dosed IV
Use is based on Fader paper. See uterine part 3 episode to review use in uterine cancer
AE/unique toxicties
Cardiotoxicity
Infusion reactions
Management/monitoring for toxicities
Cardiotoxicity: pretreatment echocardiogram, repeat every 3 months during treatment and at completion of treatment
Risk higher in patients who previously received anthracycline chemo or chest wall radiation
Hold treatment if there is a reduction in LVEF ≥ 16% from baseline, or if the LVEF is below normal limits and has dropped ≥10% from baseline; if treatment is held for at least 4 weeks and LVEF returns to normal, trastuzumab can be restarted.
Infusion reactions: If a patient experiences anaphylaxis, angioedema, interstitial pneumonitis, or ARDS, treatment should be discontinued and if the reaction is severe, that warrants permanent discontinuation.
Antibody-Drug Conjugates (ADCs):
ADCs include a targeted monoclonal antibody (like HER2), which is fused by a linker to a small cytotoxic molecule, which is known as the payload. Dosed via IV.
Trastuzumab-Deruxtecan (brand name Enhertu): HER2-directed ADC
FDA approval: unresectable/metastatic HER2 positive solid tumors (IHC 3+) (tumor site agnostic)
Gastric cancer IHC cutoffs
Based on DESTINY trials
Deruxtecan = payload = topoisomerase inhibitor
Starting dose: 5.4 mg/kg every 3 weeks, dose reductions available
Common AE: GI (nausea, vomiting, diarrhea, constipation, stomatitis, abdominal pain, dyspepsia); cardiotoxicity (LV dysfunction), fatigue, edema MSK pain, dermatologic (alopecia, rash), respiratory symptoms (URI, cough, pneumonitis, ILD, dyspnea, headaches), cytopenias
Management/monitoring for AE
Emesis: prophylactic antiemetics
Pneumonitis: consider a baseline SpO2, PFTs, and high-resolution CT. Consider repeating the high-resolution CT every 12 weeks, and more frequently if respiratory symptoms are present
Treatment should be interrupted even with grade 1 pneumonitis, and if patients have grade 2 signs or symptoms, treatment should be permanently discontinued and treatment with steroids initiated
LV dysfunction:
treatment should be interrupted if LVEF gets below 45% and has decreased 10-20% from baseline
if LV function recovers to within 10% of baseline, treatment can be resumed, but if LVEF gets below 40% or change from baseline is >20%, treatment should be discontinued permanently.
Cytopenias: see drug website for management recommendations
Mirvetuximab soravtansine (aka MIRV, brand name Elahere)
ADC against folate-receptor-alpha, which is a transmembrane glycoprotein encoded by the FOLR1 gene
FRa has limited expression in normal tissues, elevated expression in ovarian, endometrial and other cancers. Fra overexpression is associated with platinum resistance.
Soravtansine = payload = microtubule disrupting agent
FDA approval: restricted to use in patients with recurrent ovarian cancer with Fra expression ≥75% with moderate to strong staining
NCCN guidelines: also recommend use, in combination with bevacizumab, for patients with Fra expression ≥25%.
Based on MIRASOL, FORWARD I, FORWARD II, SORAYA trials
Dosing: IV, starting dose 6mg/kg (adjusted ideal body weight). Dose reductions available
Unique toxicities
Ocular: blurred vision, keratopathy, dry eye. 11% of patients experience a grade 3+ event
Median time to onset: 5.5w. Only 50% of people with this will have complete resolution.
Pneumonitis
Peripheral neuropathy
Hepatic toxicity
Managing/monitoring AE
Premedicate: IV corticosteroid, antihistamine, antipyretic, antiemetic
Ocular: prevent with use of lubricating and ophthalmic topical steroid eye drops; eye exam prior to treatment initiation and throughout treatment.
Cannot wear contact lenses during treatment!
Consult drug prescribing website for specific management instructions
Hepatic: avoid in patients with moderate to severe hepatic impairment
Tisotumab vedotin (brand name Tivdak)
ADC against tissue factor. TF is expressed on the surface of many tumor cell and is useful for tumor related angiogenesis, thrombogenicity, tumor growth, metastasis.
Vedotin = payload = Monomethyl auristatin E (MMAE), a microtubule disruptor
FDA approval: recurrent or metastatic cervical cancer
Based on InnovaTV study series
Dosing IV, starting dose 2mg/kg q3weeks. Dose reductions available.
Common AE: bleeding/hemorrhagic events (epistaxis, vaginal bleednig), GI toxicity (Nausea, diarrhea/constipation, decreased appetite), ocular (conjunctivitis, corneal toxicity, dry eye), peripheral neuropathy, epistaxis, alopecia, fatigue, anemia
Treatment discontinuation occurred in 15% of experimental arm patients in the study
Management/monitoring of toxicities: depends on grade
Ocular: involve an eye specialist. Often treatment can be held and restarted, but refer to prescribing info for details
Prevention: ophthalmic exam before initiating, throughout treatment, avoid contacts, and use eye drops (steroid, vasoconstriction, lubricating all required), cold packs during treatment
Grade 3+ peripheral neuropathy: permanent discontinuation
Any pulmonary/CNS hemorrhagic events or grade 3+ hemorrhagic events in other organs: permanent discontinuation
Grade 3+ pneumonitis: permanent discontinuation
Grade 3+ severe cutaneous reaction: permanent discontinuation
MEK inhibitors
Trametinib: MEK1 and MEK2 inhibitors; inhibit MAPK pathway
Evaluated in GOG 281, LGSOC
Dosed orally (whole tablets, empty stomach, has to be stored in the fridge!)
2mg daily starting dose. Reductions available
70% of patients required at least one dose reduction, >⅓ discontinued
Common AE:
fatigue, skin rash, anemia, GI AE
Hypertension
Retinal toxicity (2% risk of retinal detachment)
Decreased EF (8% of patients)
Management/monitoring of AE
Consider prophylactic clindamycin cream or acne medication to prevent rashes
Cardiac: echo/cardiac eval prior to initiating
NTRK fusion mutation targeting drugs
NTRK fusions occur in ~1% of all cancers
Larotrectinib: tropomysin receptor kinase inhibitor: blocks downstream signaling of the RAS/MAPK and PI3K/AKT pathways
FDA approval: tumor agnostic for tumors with NTRK gene fusion on molecular testing (from NAVIGATE trial)
Dose: orally, 100mg BID
Unique toxiciites: CNS effects (cognitive impairment, dizziness, mood disorders), skeletal fractures, hepatoxocities
Management/monitoring of AE
Hepatoxicity: if patients have moderate/severe hepatic impairment, start at 50% dose
Entrecitnib: TKI, multikinase (NTRK, ROS1, ALK fusion)
FDA approval: disease agnostic FDA approval for patients with NTRK fusion mutations. In lung cancer, it’s also approved for ROS1 mutations (from STARTRK-2 trial)
Dose: orally, 600mg daily
Has better CNS penetration than larotrectinib (may be a better choice for patients with CNS metastases)
Toxicities, severe/unique: CHF, hyperuricemia, CNS effects (see larotrectinib above), skeletal fractures, hepatotoxicity, CYP3A substrate
Management/monitoring of AE
Cardiac: cardiac eval before and during treatment
Hyperuricemia: monitor uric acid levels